Bovine Herpes Virus Type 1 Vaccine
In Stock
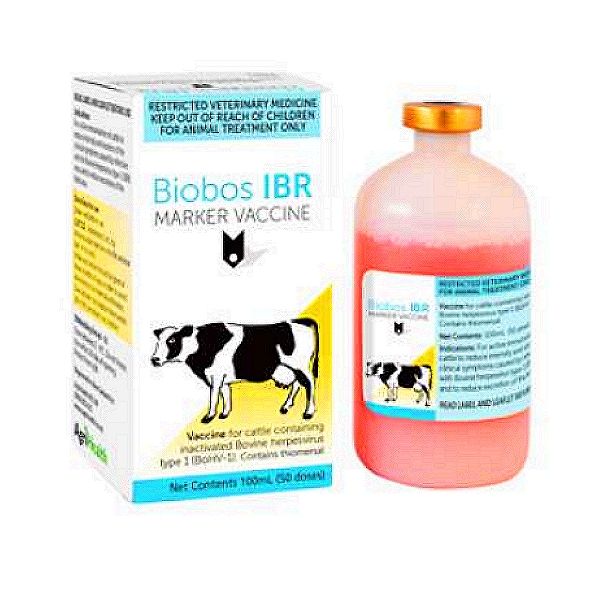
Bovilis IBR Marker Live Vaccine
AdriHealth BioBos IBR Marker vaccine is indicated for the active immunization of cattle against conjunctivitis and vulvovaginitis caused by Bovine Herpes Virus Type 1. Enables vets to distinguish between animals that have been vaccinated, and those exposed to the wild-type virus through serological testing. A WVA Veterinary Medicine.Categories: Antivirals, B, Vaccines